Science Has Found the Exact Sleep Window That Slows Your Biological Aging — And Most People Miss It
Most adults know they should be sleeping more. But a landmark study published in Nature in May 2026 reveals that the conversation about sleep needs to go much deeper than counting hours. A team of researchers at Columbia University has demonstrated, for the first time at this scale, that sleep duration affects the biological age of 17 different organ systems simultaneously — and that the optimal window for slowing whole-body aging is surprisingly precise: between 6.4 and 7.8 hours per night. Just as striking: both sleeping too little and sleeping too much accelerate biological aging. Understanding this curve, and landing consistently within its optimal range, may be one of the highest-leverage changes any person can make for their long-term health.
Why Sleep Is as Essential as Food and Water for Long-Term Health
The underappreciated role of sleep in whole-body repair
Sleep is commonly framed as a brain phenomenon — rest for an overworked mind. But the biology tells a far more comprehensive story. During sleep, the body runs a coordinated, whole-system maintenance program that cannot be replicated in wakefulness. The immune system releases cytokines that regulate inflammation and fight infection. Growth hormone surges during deep slow-wave sleep, driving cellular repair and tissue regeneration throughout the body. Cortisol drops to its daily low, giving the adrenal system time to reset. The liver metabolizes compounds it cannot process effectively during waking hours. And in the brain, the glymphatic system — a waste-clearance network that operates primarily during sleep — flushes toxic proteins including amyloid-beta and tau, the proteins implicated in Alzheimer's disease.
This is not rest. This is active, essential biological work that only happens during sleep — and it spans every major organ system in the body.
How modern life is eroding our sleep
Average sleep duration has declined significantly over the past century, driven by artificial light, screen use, shift work, extended commuting, and a cultural ethos that treats sleep as optional. The average adult now sleeps roughly 6.8 hours per night — already at the lower edge of the optimal range identified in this new research. Large proportions of the working-age population consistently sleep fewer than 6 hours. Understanding the biological consequences of this erosion — measured across 17 organ systems, not just the brain — gives this research an urgency that earlier sleep studies could not match.
The Half-Million-Person Study That Redefined Optimal Sleep
Who conducted it and why it matters
The study was led by Dr. Junhao Wen, assistant professor of radiology at Columbia University Vagelos College of Physicians and Surgeons. His team's motivation came from a gap in prior research: although sleep had been linked to brain aging and mental health outcomes for decades, its effects on the rest of the body's organs had never been systematically measured with biological aging clocks at this scale. The paper, titled "Sleep chart of biological ageing clocks in middle and late life," was published in Nature on May 13, 2026, and represents one of the most comprehensive analyses of sleep and whole-body aging ever conducted.
The UK Biobank — scope and data sources
The study drew on data from the UK Biobank, one of the largest and most detailed biomedical databases in the world, with approximately 500,000 participants ranging in age from 37 to 84 years. Three distinct categories of biological data were used to construct aging clocks:
- Structural MRI imaging: providing organ-specific morphological data for brain, heart, liver, and other tissues
- Plasma proteomics: blood protein profiles that reflect organ-specific biological activity
- Plasma metabolomics: blood metabolite profiles that capture metabolic function across systems
Sleep duration was obtained from participant self-reports. This multi-layer data approach allowed researchers to build and cross-validate aging clocks from multiple biological angles within the same individuals.
Why this study is different from prior sleep research
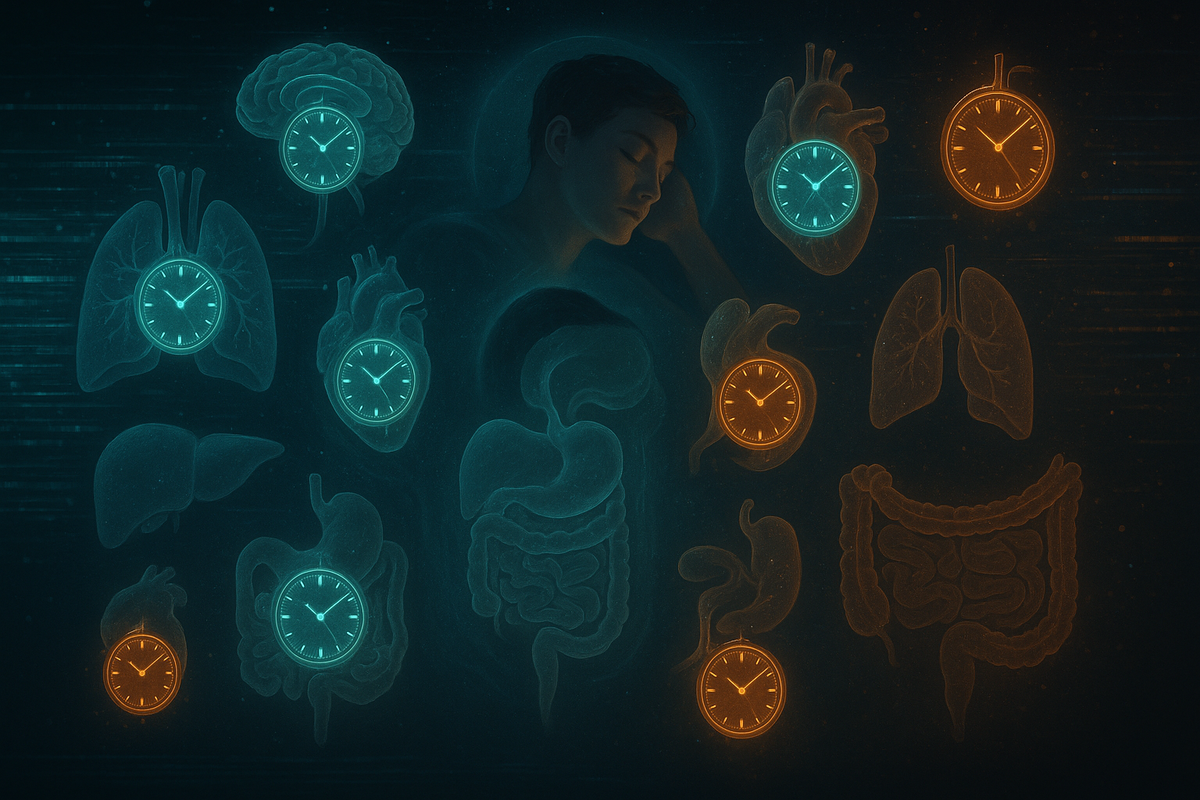
What distinguishes this study is its scope and methodology. Rather than assessing a single biomarker or one organ system, the team constructed 23 separate aging clocks across 17 organ systems — including the brain, heart, lung, liver, kidney, immune system, digestive system, adipose tissue, and musculoskeletal system. Prior sleep research typically found associations between sleep and specific outcomes in isolation. This study provides a panoramic, simultaneous view of how sleep affects biological aging across the entire body.
Biological Aging Clocks — How Scientists Measured Sleep's True Impact on Organs
What is a biological aging clock?
A biological aging clock is a machine-learning model trained on biological data — proteins, metabolites, or imaging features — that can estimate how old a particular organ is biologically, independently of a person's chronological age. Think of it like an odometer for your liver or your heart: one that reads off biological wear rather than calendar years. The key measure is the biological age gap — the difference between an organ's estimated biological age and the person's actual age. A positive gap means the organ is aging faster than expected; a negative gap means it is aging more slowly.
How 23 clocks were built for 17 organs
For each organ system included in the study, multiple aging clocks were built from different data layers. For the liver, the team built a protein-based aging clock, a metabolomics-based aging clock, and an imaging-based aging clock — allowing them to detect aging signals from multiple biological angles simultaneously. This multi-omics approach ensures the findings are not artifacts of a single measurement method. Across the full study, 23 clocks covered 17 organ systems: nine brain systems, as well as heart, lung, liver, kidney, immune, adipose, digestive, and musculoskeletal systems.
Why organ-specific clocks reveal more than whole-body measures
Organs age at meaningfully different rates. Research has long shown that ovaries, for example, age faster than most other organs — a fact with significant implications for fertility. The brain's aging trajectory differs from the heart's. The liver has its own metabolic aging clock that can accelerate independently of systemic aging. By building organ-specific clocks rather than relying on a single whole-body measure, the researchers could identify which organs are most affected by sleep disruption, and how the mechanisms differ between under-sleeping and over-sleeping.
The 6.4–7.8 Hour Sweet Spot Associated With Slower Aging and Greater Longevity
When the team assessed the relationship between sleep duration and biological age gaps across all 23 clocks, a consistent pattern emerged: a U-shaped curve. At the bottom of the U — the point associated with the least biological age acceleration — was a sleep duration range of 6.4 to 7.8 hours per night. People consistently sleeping within this range showed slower biological aging across nearly every organ system measured.
The exact optimal point varied slightly by organ and by sex, reflecting the biological complexity of aging. The brain's optimal sleep duration may differ slightly from the liver's, and women's optimal range may be slightly different from men's — though both remain within the general 6.4–7.8 hour window. There is no single magic number, but rather a zone of optimal sleep that most adults should be aiming for.
It is worth noting that this study, like all population-level research, demonstrates association rather than definitive causation. A person who is chronically ill may sleep poorly, and that poor sleep may then further accelerate aging — creating a reinforcing loop. The researchers used Mendelian randomization analysis to address reverse causality, and while it did not provide definitive proof that sleep duration causally drives aging, it did not support the alternative explanation either.
How Too Little Sleep Speeds Aging in Your Brain, Heart, and Lungs
Short sleep — fewer than 6 hours per night — was associated with accelerated aging across most organ systems, with particularly pronounced effects in the brain, cardiovascular system, and respiratory system. Diseases specifically linked to short sleep duration in this dataset included depression, anxiety, obesity, type 2 diabetes, hypertension, ischemic heart disease, and heart arrhythmias.
Brain and mental health aging
Short sleep disrupts the brain's two primary maintenance functions: memory consolidation (the process by which the hippocampus transfers experiences into long-term memory during slow-wave and REM sleep) and glymphatic clearance (the overnight flushing of toxic proteins including amyloid-beta and tau). Chronic insufficient sleep has been shown to increase amyloid burden in the brain — a direct precursor to Alzheimer's disease pathology. Beyond dementia risk, short sleep directly and strongly predicts depression and anxiety, with the brain's aging clock showing significant acceleration in short sleepers.
Cardiovascular aging
During normal sleep, blood pressure drops by 10–20% in what researchers call "nocturnal dipping" — a restorative process for the cardiovascular system. Short sleep disrupts this dipping pattern, keeping the heart and blood vessels under elevated pressure for more hours per day. Combined with elevated overnight cortisol and increased sympathetic nervous system activity in short sleepers, the result is a measurably accelerated cardiovascular aging clock, with elevated rates of hypertension, ischemic heart disease, and arrhythmias.
Respiratory and metabolic aging
Short sleep drives systemic low-grade inflammation through elevated pro-inflammatory cytokines. This inflammatory state is directly linked to asthma and COPD exacerbation, as well as insulin resistance — a key driver of type 2 diabetes and obesity. The metabolic aging clock in the liver and adipose tissue shows clear acceleration in habitual short sleepers.
Why Too Much Sleep Is Also Linked to Faster Aging and Higher Disease Risk
Here is where the research surprises many readers: sleeping significantly more than 7.8 hours per night is also associated with accelerated biological aging across most organ systems. The U-shape is real and symmetrical.
What drives the long-sleep aging effect?
Researchers emphasize that long sleep is often more of a symptom than a cause. Chronic disease, depression, high levels of physical inactivity, and metabolic dysfunction are all conditions that cause people to sleep longer — and these same conditions accelerate biological aging independently. The adipose tissue aging clock shows a particularly strong association with long sleep, linking excessive sleep to inflammatory fat tissue pathways. Extended time in bed also disrupts circadian rhythm integrity: lying in bed awake for long periods fragments sleep architecture and reduces the quality of slow-wave and REM sleep even when total time in bed is long.
Different pathways for long vs. short sleepers
One of the most clinically significant findings from the mediation analysis is that long and short sleep reach some of the same health outcomes — particularly late-life depression — via different biological pathways. Short sleep acts on depression through a more direct mechanism. Long sleep influences depression via the brain and adipose aging clocks as intermediate steps. This distinction matters: it suggests that long sleepers and short sleepers should not receive identical clinical interventions.
Diseases Strongly Linked to Both Short and Long Sleep Duration
Across both ends of the sleep duration spectrum, the UK Biobank survival analysis identified consistent disease associations:
- Brain and mental health: depression and anxiety disorders (most strongly linked to short sleep, but also elevated with long sleep)
- Cardiovascular: hypertension, ischemic heart disease, cardiac arrhythmias (primarily short sleep)
- Respiratory: COPD and asthma (both short and long sleep)
- Digestive: gastritis and gastroesophageal reflux disease / GERD (both short and long sleep)
- Metabolic: obesity and type 2 diabetes (primarily short sleep)
- Immune function: broad immune dysregulation and increased susceptibility to infection (both short and long sleep)
The study used time-to-incident disease predictions — tracking when participants actually developed these conditions over follow-up time — giving the disease associations stronger longitudinal credibility than cross-sectional studies.
Is Sleep Duration Mostly Genetic, or Can You Change It?
The genetic component of sleep duration
Sleep duration has a meaningful genetic component. Chronotype — whether you are naturally a morning person or a night owl — is substantially inherited, as are baseline sleep need and sleep architecture. The study's Mendelian randomization analysis used genetic variants associated with sleep duration as natural instruments to probe causation, finding that genetic evidence did not strongly support the idea that disease causes sleep duration changes — but could not completely rule it out.
What this means practically: genetics explains some of the variation in sleep patterns between individuals, but not all of it. Studies of identical twins consistently show that a substantial portion of sleep duration variation is environmental and behavioral — and therefore modifiable.
How much can behavior shift your sleep duration?
Cognitive Behavioral Therapy for Insomnia (CBT-I) is the most evidence-supported intervention for improving both sleep duration and quality, consistently outperforming sleep medication in clinical trials with more durable results and no dependency risk. Beyond CBT-I, consistent wake times, morning light exposure, reduced alcohol intake, regular aerobic exercise, and cool bedroom temperatures all reliably shift sleep duration toward the optimal range for most adults.
Could Disrupted Sleep Be an Early Warning Sign of Undiagnosed Disease?
If you are consistently sleeping fewer than 6 hours or more than 9 hours without intending to — without an alarm forcing you up or a deliberate lifestyle choice — it may be worth treating this as a potential clinical signal rather than just a lifestyle issue.
Sleep apnea, which affects an estimated 1 billion people worldwide and remains massively underdiagnosed, is one of the most common drivers of short and fragmented sleep. Its symptoms — snoring, gasping, non-restorative sleep, morning headaches, daytime fatigue — are often dismissed as normal. Untreated sleep apnea independently accelerates the cardiovascular, metabolic, and cognitive aging clocks identified in this study.
Depression is both a cause and a consequence of disrupted sleep. Thyroid dysfunction, chronic pain, and cardiovascular disease all alter sleep patterns before they are diagnosed. If sleep hygiene interventions do not improve chronic sleep duration within a few weeks, consultation with a healthcare provider is warranted.
Science-Backed Strategies to Optimize Your Sleep for Healthy Aging
Translating the research into practice means working toward consistent sleep within the 6.4–7.8 hour window. Here are eight of the most evidence-supported strategies:
- Set a consistent wake time every day, including weekends. This is the single most powerful anchor of the circadian clock, stabilizing sleep pressure over time.
- Limit total time in bed to your actual sleep need. If you are consistently lying awake for long periods, slightly restricting time in bed builds sleep pressure and improves sleep efficiency.
- Get morning bright light within 30 minutes of waking. Natural sunlight or a 10,000-lux light therapy lamp resets the circadian clock's daily start signal.
- Reduce blue light exposure in the 2 hours before bed. Blue-wavelength light suppresses melatonin; blue-light glasses or screen filters help reduce this effect.
- Keep your bedroom cool — between 18–20°C (65–68°F). Core body temperature needs to drop for sleep onset; a cool room facilitates this critical shift.
- Limit alcohol, especially in the 3 hours before bed. Alcohol induces sleep but suppresses REM sleep and causes early-morning wake-ups, reducing both sleep quality and effective duration.
- Exercise regularly, but avoid vigorous exercise within 2–3 hours of bedtime. Aerobic exercise consistently improves both sleep duration and deep sleep quality when done earlier in the day.
- If good sleep hygiene does not produce 6+ hours of restorative sleep, consider a sleep study. Sleep apnea and other sleep disorders cannot be resolved by lifestyle changes alone.
Start with one change this week. Even a single behavioral shift — a consistent wake time, morning light, reduced evening alcohol — can meaningfully improve sleep duration over 2–3 weeks of consistent practice.
Conclusion
The science of sleep just got significantly more precise. A half-million-person study, analyzing 23 biological aging clocks across 17 organ systems, has given us the clearest picture yet of how sleep duration affects how fast — or slowly — the human body ages. The 6.4 to 7.8 hour window is not an arbitrary guideline: it is the range associated with the least acceleration of biological aging across the brain, heart, lungs, liver, immune system, and beyond.
The takeaway is not that you need to be perfect every night. It is that consistently landing within this window — and avoiding both chronic sleep deprivation and habitual excessive sleep — is one of the most powerful, accessible tools available for slowing biological aging. Unlike genetics, unlike pharmaceutical interventions, sleep is something most people can meaningfully improve within weeks. This research is an invitation to take sleep as seriously as diet and exercise.